Xiaoliang Sunney Xie became the first tenured professor (1998) as well as the first endowed chair professor (2009) at Harvard University among scholars of mainland China origin since the 1978 Chinese Economic Reform. He is a member of the United States National Academy of Sciences and National Academy of Medicine. He received the Peter Debye Award of American Chemical Society and Founder's Award of Biophysical Society, the highest honors for physical chemistry and biophysics in the US, respectively, and was the first Chinese American to receive the Albany Prize in Medicine and Biomedical Research.
Xiaoliang Sunney Xie
In July 2018, Xie returned to Peking University, his alma mater, as the Lee Shau-kee Professor and Director of the Biomedical Pioneering Innovation Center (BIOPIC) and Beijing Innovation Center for Genomics (ICG).
As a tribute to the University's 120th anniversary in 2018, he narrated his lifelong love affair with PKU. Today, we share with you his fascinating story.
As if barely a twinkle in time, shining immortal down history's passage, it was a century and one score years ago when our alma mater, Peking University, was born.
A nation’s rebirth and the making of a scientist
With my parents and younger brother in front of the new Peking University Library, 1975
I was born and bred in Beida, the rightly affectionate name for PKU, where its views and vistas, each bit and every cranny, are the most dear to me. From Beida's affiliated kindergarten, primary and middle schools, then onward to the college, most of my formative years took place on this very campus, Beida and I braved through the tides of time and fortune together, shaping bonds that could never be undone. Each return upon its soils brings a warmth which assures and soothes.
Beida is not just my school, but also my home; a temple for knowledge, and the anchor of my heart. After spending more than three decades in the United States, I choose now to return to Beida, to pick up again my romance with this place: once the cradle of my beginnings, now the crest of my life's work.
I was born in 1962 at Peking University's picturesque Langrun Place. My father, Xie Youchang, and my mother, Yang Junying, were then both college teachers of chemistry at PKU. My childhood home was filled with books, and our days were peaceful and happy. The Beida campus, also known as Yen Garden, is possibly the most beautiful college campus in the world. It was once the site of Yenching University, and part of the imperial gardens of both Ming and Qing Dynasties.
Yen Garden was my childhood paradise. In Spring, a thousand flowers bloomed; come Summer, the grounds were lush and bustling of life. As a boy I would capture dragonflies by the Weiming Lake, then set them free, observing their dancing wings up in the azure blue sky.
A childhood photo taken by Weiming Lake
Autumn was the most beautiful season in Yen Garden. Ginkgo leaves, stained a lovely yellow, fluttered gorgeously against the red walls and green tiles of campus buildings. In the winter, Weiming Lake became an ice wonderland, with children on skates gliding and twirling across its mirror-like surface.
That idyllic picture was shattered in 1966. The "Cultural Revolution" had begun, and all activities were suspended in the schools. My parents, as university teachers, were thrown into the whirls of political movements. I would never forget, in the dead of the night, when Red Guards marched into the houses of my neighbors in Langrun Place to confiscate their property, abuse and insult them, and take them away. As a young child I cowered in a corner with fear, not understanding but knowing that everything had changed. Cut off from teaching and research, Father was deported to the May Seventh Cadre School in Jiangxi Province for "labor education", while my mother, younger brother and I were to stay in Beijing, hundreds of miles apart.
Against the backdrop of the "Cultural Revolution" at its height, a child's world carried on with innocence. I remember that it was 1969, as I first entered elementary school, when Father returned to Beijing. Having learned the crafts of masonry and carpentry in Jiangxi, he made a spinning top especially for me. This exquisitely crafted, endlessly spinning toy sparked my curiosity.
The first woodwork I crafted using Father's toolbox was a level scale – this was the first precision measurement tool I designed in my life! The hobby proved to be irresistible, and I began making model airplanes and ships, and once even a working speaker. My hands got more adroit as my craft became ever more complex, and my curiosity about the science behind my creations grew stronger and stronger.
In middle school, I began making various electronic equipment, amongst them a superheterodyne receiver, remote-controlled model ships, and a set of loudspeakers. My interest in experimental science thus began in electronics, and my calling in life was revealed to me – I would become a scientist.
The National College Entrance Examination was reinstated during my high school years. Life at the Peking University Affiliated High School had gone back to normal, our study environment was one of keenness and camaraderie. Besides formal education and prepping for the college entrance exam, we also enjoyed many extracurricular activities. I was the class monitor and an ace spiker of our class volleyball team. Many of my classmates were children of Beida staff and faculty, we indulged our talents and passions in diverse pursuits. I remember how my classmate and friend Yu Lian produced a beautifully penned futuristic radio drama that was very well-received by our fellow classmates. I had by then grown fond of classical music, immersing myself in the feast for the ears, and also obsessed with inventing speakers with improved acoustics.
As a high school student, I once wrote an essay entitled "Old Summer Palace (Yuanming Yuan)". I employed the scenery in Yuanming Yuan, an architectural wonder once called "the Garden of Gardens" that was reduce to rubble by British and French Imperial forces in the final turbulent years of the Qing Dynasty, as a metaphor for the ruinations left by the Cultural Revolution, and looked forward to the Chinese economic reform, which began in 1978, as the country's hope for rebirth. For its apt imagery and patriotic sentiment, my essay was chosen by our Chinese teacher to be passed and read in class.
With my Peking University Affiliated High School classmates in the Old Summer Palace (Yuanming Yuan), 1978
Since my schooling days, I had always preferred to be in charge of finding my own projects, be it penning essays, or crafting by hand. The greater the significance of the project, the greater its challenges, the more joy it would bring me by completion. I still remember that, when Professor Wang Xuan of the PKU Institute of Computer Science and Technology was leading the development of laser typesetting systems for the Chinese language, I and other students of the University's Affiliated Primary School helped to manually input digitized fonts one by one. Many years later, as we were awed by the world's first Chinese Language Laser Typesetting technology which replaced traditional typesetting, the pride brought by my role in the project, albeit only as a young volunteer, convinced me further that I too should be aiming big. Later on, "aiming big" would become the motif of my career as a scientist.
My parents were finally able to return to their beloved teaching and research during my high school years. Father would write a college textbook for Structural Chemistry, while consumed in his research and constantly delighting himself with breakthroughs. Mother devoted herself to teaching, and was deeply loved and respected by her students. By their examples, I too felt the attraction in teaching and research. After graduating from high school, I was admitted to Peking University, accepted by my first choice, the Department of Chemistry.
Temple of knowledge and the sprout of innovation
In 1980, I began life as a Beida undergrad, taking with me my childhood dreams, hopes for the future, and a thirst for knowledge.
Since the May Fourth Movement in 1919, Peking University has been a beacon for the ideals of democracy, science, and patriotism. Beida students in the early 1980s, driven by a strong sense of national responsibility, eagerly pursued and aspired to democracy and progress. All sorts of philosophies and schools of thought flourished on campus, and a three-sided concrete lawn called "The Triangle", the heart of student life where events and ideas could be demonstrated on posters and exhibits, became entrenched in the memory of Beida scholars from those bygone days.
Peking University was above all a center of learning, with scholarship both its cornerstone and lodestar. Most Beida students believed in modern science as a cure for the many ails of our ancient country. Having aspired to be a scientist since high school, I immediately immersed myself in learning and discovery after entering the land of academic freedom that was Beida.
As an undergraduate student of Peking University
Beida set me free to explore an ocean of knowledge. I audited classes from other departments out of interest: classical mechanics, quantum mechanics, statistical mechanics and electrodynamics from the Department of Physics, classes in electronics from the Department of Electronic Engineering, statistics classes in the Math Department, and so on. I would be richly rewarded for these efforts.
My high school classmate Yu Lian entered the PKU Department of Chemistry together with me. After class, we often conducted academic discourses, explored unanswered questions, and encouraged each other. Lian is currently a professor at the University of Wisconsin-Madison School of Pharmacy.
Graduation picture taken in 1984 with Yu Lian (right)
During the summer of my freshman year, I taught myself computer programming, and was often using the computers in the Beida North Pavilion. After much thinking, I found that the energy in ionic crystal is an infinite series, which requires a large amount of calculation, and I attempted to write a program in Fortran to calculate the energy of crystal structures. This project may seem insignificant to readers of today, but that was the first time that I, a Chemistry major, was able to use computer programming to solve such an "interdisciplinary" problem, and the experience was intensely gratifying.
Besides concentrating on my studies, I remained a huge fan of volleyball. My favorite team, the China men's national volleyball team, qualified for the World Cup against all odds my sophomore year in 1981. Deeply inspirited by the victory, Beida student shouted the slogan of "Let's united and revitalize China" across campus, the echoes of which soon spread to the rest of the nation. In the following years the China women's national volleyball team would go on to win the Volleyball World Cup, World Championship, and an Olympic gold medal. Such triumphs in the sporting world heartened and inspired the young men and women of Peking University, and those precious memories of student life would always be dear to my heart.
In my senior year I had the good fortune to do my graduation thesis under Professor Cai Shengmin of the Department of Chemistry, in Beida's South Chemistry Hall. Professor Cai was an electrochemist and a superb experimentalist. He had a wide range of interests, a lively intellect, great sense of humor, was always full of energy, and spoke fluent English.
His personal and academic guidance to me would have a profound influence on my career. Professor Cai was good at using vivid language to illustrate complex and abstract concepts. My thesis was on computer-controlled photoelectrochemical reactions, which involved lock-in amplifiers. I could still recall clearly how Prof. Cai explained the working principle of lock-in amplifiers. In the process of working on my thesis I began to realize that innovations in scientific instrument can often lead to breakthroughs in scientific research. This would be proven further in my own scientific career.
Reunion with my college mentor Professor Cai Shengming of Peking University (left) when I was studying in the States
College is the time for the accumulation of knowledge, but research is not accumulation but rather the creation of new knowledge. The biggest challenge for researchers is to develop and maintain their ability to innovate. My childhood, youth and college years spent in Beida had nurtured in me the sprout of innovation, and made science my life's true calling.
After getting my bachelor's degree, I stayed in Beida for one more year as a master student. In those days, China was very much behind the rest of the world in science and technology, and I decided to go overseas for further study.
My generation was much more fortunate than that of our parents. The Chinese Economic Reform made it possible for me and many of my classmates to study abroad. The year I graduated, students of Peking University held a banner reading "Hello, Xiaoping" during a parade on Tiananmen Square celebrating the 35th anniversary of the People's Republic's founding. It was the salutation from the bottom of our hearts.
Breakthroughs and impacts
In 1985, at the age of 23, I bid farewell to Peking University for the first time and left for America, beginning a new chapter in my life. I did my Ph.D. in chemical kinetics at the University of California, San Diego under Professor John Simon, studying ultrafast chemical reactions with ultrashort picosecond (10-12 seconds) laser pulses. With Professor Simon's strong support, I successfully realized my idea of studying conformational dynamics of biological macromolecules by time-resolved circular dichroism, which eventually became my doctoral thesis [1]. In developing this technique, I made use of the lock-in amplifier that Professor Cai Shengmin had introduced to me earlier on.
Ph.D. graduation ceremony with father at the University of California, San Diego in 1990
I went on to the University of Chicago, where I did a short postdoc in the lab of the distinguished physical chemist Professor Graham Fleming. It was there where I first embarked on an emergent research direction, which would eventually define my independent career: single-molecule spectroscopy and imaging at room temperature.
In 1992 I joined the Pacific Northwest National Laboratory (PNNL) as the first scientist originated from mainland China, and established my own research group. Soon I was able to do single molecule fluorescence imaging at room temperature. Washington state, where PPNL is located, was heavily polluted by nuclear and chemical wastes during the Cold War, which led to the US Department of Energy building a USD $250 million "Environmental Molecular Sciences Laboratory" at PNNL, hoping to address environmental problems through basic research.
In my lab at the Pacific Northwest National Laboratory
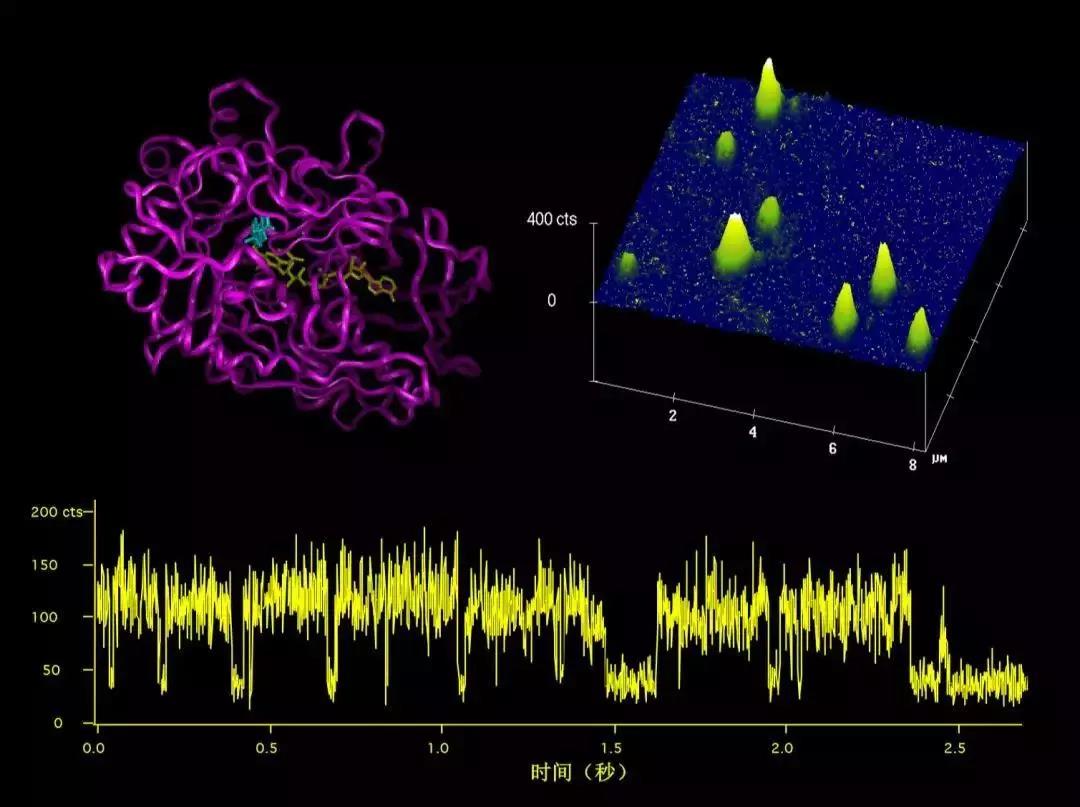
In 1998, thanks to the support we received at PNNL and previous advances made by my group in fluorescence microscopy, my postdoc Peter Lu (a fellow PKU Department of Chemistry alumnus) and I first reported in Science the real-time monitoring of stochastic enzymatic cycling of a single enzyme molecule by fluorescence detection [2]. This work proved to be groundbreaking— chemical reactions of a single molecule occurs stochastically, i.e. waiting time required for the reaction to occur is randomly distributed, instead of being predictable in conventional experiments involving a large number of molecules. Many biological macromolecules in cells, such as DNA, exist in the form of single molecules. The possibility of real-time observation of single-molecule chemical reactions would change the way many biological questions are addressed.
Fluorescence imaging (top right) of single-molecules of cholesterol oxidase (top left) obtained at PNNL in 1998 and real-time detection of stochastic enzymatic cycling of one of the enzyme molecules
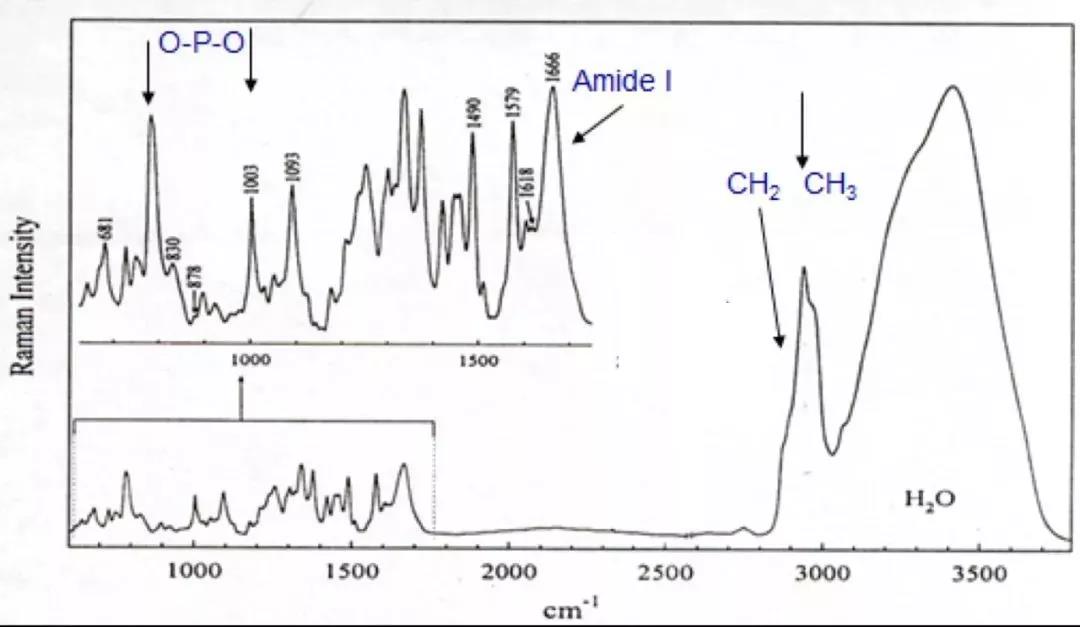
On another front, my lab also developed a label-free Raman Scattering imaging technique [3]. In 1928, the Indian scientist C. V. Raman discovered the inelastic scattering of light by molecules, a phenomenon which now bears his name and earned him the Nobel Prize in Physics. Raman spectroscopy can be used to measure the characteristic vibrational frequency of different molecules, but the signal is extremely weak and requires a long detection time. Later, progress in laser and nonlinear optics greatly enhanced the signal, but technical difficulties had limited application of Raman spectroscopy in the imaging of biological samples. Our new technique made rapid nonlinear Raman imaging in biological samples a reality.
Raman spectroscopy of the cell showing vibrational frequencies of chemical bonds characteristic of different molecules (water, lipid, protein, DNA). However, traditional Raman spectroscopy produces weak signals requiring long-term detection (>0.1 seconds for each point, 600x600 points for imaging requiring >10 hours). We eventually realized video-rate Raman imaging.
These two projects produced the most highly cited papers from my lab to date. A new door opened when, in 1998, I was offered a tenured professorship by the Department of Chemistry and Chemical Biology at Harvard University.
Next to Harvard University's Widener Library stood an exquisite stone monument from China. Known as the Harvard Bixi, it is a sculpture of a mythical dragon with the shell of a turtle, bearing a 17-foot high, 27-ton marble stele on its back. This was a gift presented to Harvard for its Tercentenary in 1936 by Dr. Hu Shih (Hu, Shi), then dean of the college of humanities of Peking University, and other Chinese Harvard alumni.
The inscription on the stele reads, "...Our nation symbolizes an ancient culture of the East, but time irrevocably moves forward and the world evolves, constantly undergoing renewal and change. Those committed to learning once again have studied overseas to deepen their knowledge and self-understanding. During the past thirty years, nearly one thousand of our countrymen have returned home upon completion of their studies at Harvard University to serve the nation and society. This is truly marvelous!”
The stone monument from Peking University, on Harvard campus [4]
Interestingly, Jim Anderson, chairman of Harvard's Department of Chemistry and Chemical Biology who recruited me, told me that his father, Paul A. Anderson, was appointed in 1925 by Yenching University President John Leighton Stuart as the first chairman of its Physics Department, and subsequently lived and worked in Yen Garden for several years [5].
Harvard's Department of Chemistry and Chemical Biology sparkles with the luster of brilliant minds. On its faculty are many leading figures in their respective fields, including four Nobel Prize winners. The rising Chinese scientist Zhuang Xiaowei also joined the department two year later, and we became close friends. In 2013, Jim, Xiaowei and I attended the 100th anniversary celebration of the Peking University School of Physics.
At the Centennial Celebration of Peking University School of Physics with Jim Anderson and Zhuang Xiaowei
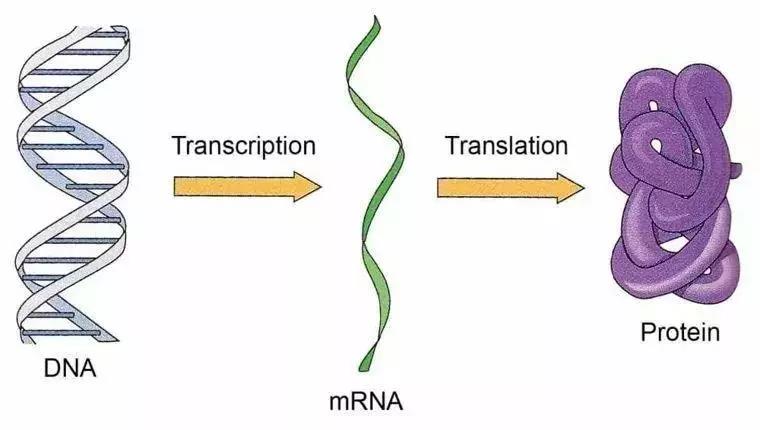
When I first arrived at Harvard, I had a hunch that the single molecule approach would have important applications in biology. Although I'd built myself a good foundation in mathematics, physics and chemistry in Beida, I never studied molecular biology, so I decided then to learn from scratch. Together with the students in my group, I audited a molecular biology course in Harvard's Department of Biology. Professor Richard Losick, who taught the class, showed cartoon movies to explain the mechanisms of biomacromolecules such as RNA polymerase and ribosomes. As I listened to the professor's lively demonstrations, my mind was already formulating how I could experimentally observe the process by which these biomacromolecules carry out gene expression. This would require visualizing the behavior of a single DNA molecule in a live cell, in which any particular gene exists in only one or two copies.
DNA exists in cells in the form of single molecules, and gene expression follows the central dogma of molecular biology
In 2006, after three years of hard work, my two "debuts" in the field of molecular biology were published simultaneously in Science and Nature. The papers reported for the first time real-time observation of protein production one molecule at a time in a bacterial cell, describing at a quantitative level the central dogma of molecular biology [6,7]. These two papers became highly influential; Professor Losick even began using real-time movies from our experiments to explain gene expression in his class. This work further reinforced the importance of the interdisciplinary approach: novel methods and tools in physics and chemistry often bring new insights and discoveries in biology, while understanding life's processes requires both quantitative experimentation and theoretical analysis.
One week after the publication of our two papers, I received a call from the Gates Foundation inviting me to apply for a grant, as they hoped our new technology might be useful in understanding why a small subgroup of tuberculosis-causing bacteria cells was showing drug resistance – tuberculosis was claiming the lives of millions of children in Africa each year. In the following year, Bill Gates, "the most successful college dropout", was awarded an honorary doctorate by Harvard and gave a very moving speech at the graduation ceremony. Later on when he visited my lab, I found that he had a deep understanding of molecular biology – I reckoned he too must have learned it on his own. What was unexpected was his familiarity with the ultrafast lasers used in our experiments. Though we have yet to solve the problem of drug resistance in those bacteria cells, the Gates Foundation project made me wonder: could the advances we make in fundamental science be used to benefit society?
In 2010, Bill Gates (right) visited my lab at Harvard, discussing science with me and my students.
Scientific research requires curiosity, inspiration, and above all ceaseless accumulation. Choosing a direction is critical – the difficulty lies not just in deciding what to do, but also what not to do. It is not easy to ask the most compelling scientific question ahead of everyone else, the choice is difficult, and often risky, requiring abundant funding and an excellent team. Whether in fundamental research or technology development, I believe the biggest challenge for the leader of a research group is to find the most significant questions, then leading the way to solving them. However, many people prefer to vaunt the impacts of their results which, in reality may only carry meager significance, instead of putting their efforts toward asking the right questions in the first place.
Lab meeting at Harvard
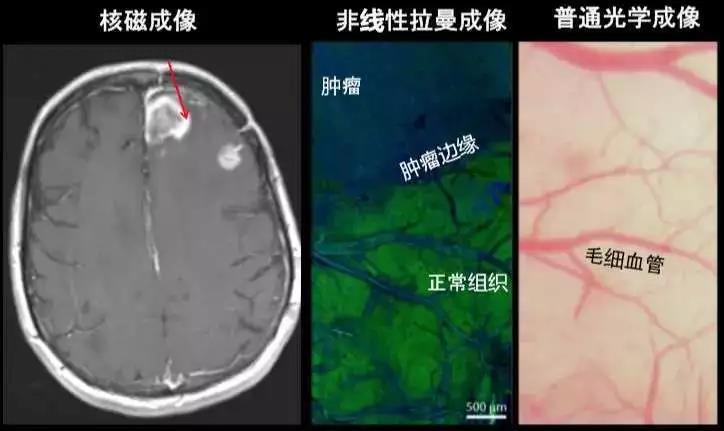
Our first effort to put our findings into practical application was employing label-free coherent Raman scattering (CRS) imaging [3,8] in distinguishing tumor margins during brain tumor surgery [9]. Magnetic Resonance Imaging allows us to visualize the location of brain tumors, but is not able to differentiate tumor cells from normal cells due to limited spatial resolution. Neurosurgery requires high-resolution optical microscopy, via the conventional hematoxylin and eosin (H&E)-stained cryosection approach, which is time-consuming and labor-intensive. Our rapid Raman imaging technology allows for a nondestructive, label-free optical method to observe tumor cells, greatly enhancing the visualization of tumor margins during surgery, and has now been commercialized and applied in neurosurgeries.
Magnetic Resonance Imaging (left) can visualize tumors in the brain, but its spatial resolution is not sufficient to distinguish tumor margins. Traditional optical imaging requires complex staining, otherwise individual cells cannot be detected (right): using label-free Raman imaging (middle), neurosurgeons can differentiate between tumors (blue, protein) and normal brain tissue (green, lipid).
At the same time, revolution in a new generation of sequencers had led to a significant drop in the cost of DNA sequencing, heralding the advent of personalized medicine. I realized the tremendous potential of pursuing this new direction, which just so happened to be in line with our forte. Thus my lab embarked upon the new venture on single-cell genomics, and we developed an original model of DNA sequencer [10] in 2011.
Speaking of transitions, any research field that was once flourishing may eventually become saturated, or even outdated, and more often than not a scientist has to explore new directions in his or her career. An experimental physical chemist requires a huge investment in laboratory equipment, and I was once worried that my changing fields might be a real challenge. I was very fortunate to have been funded twice by the NIH Director's Pioneer Award, which supports high risk research projects with the potential of high return, and allowed me to weather through the lean years during my transition period.
With Xie lab members working on single-cell DNA amplification
In 2012, we invented a single-cell DNA amplification method called MALBAC (Multiple Annealing and Looping-Based Amplification Cycles), which enables DNA sequencing of individual human cells [11].
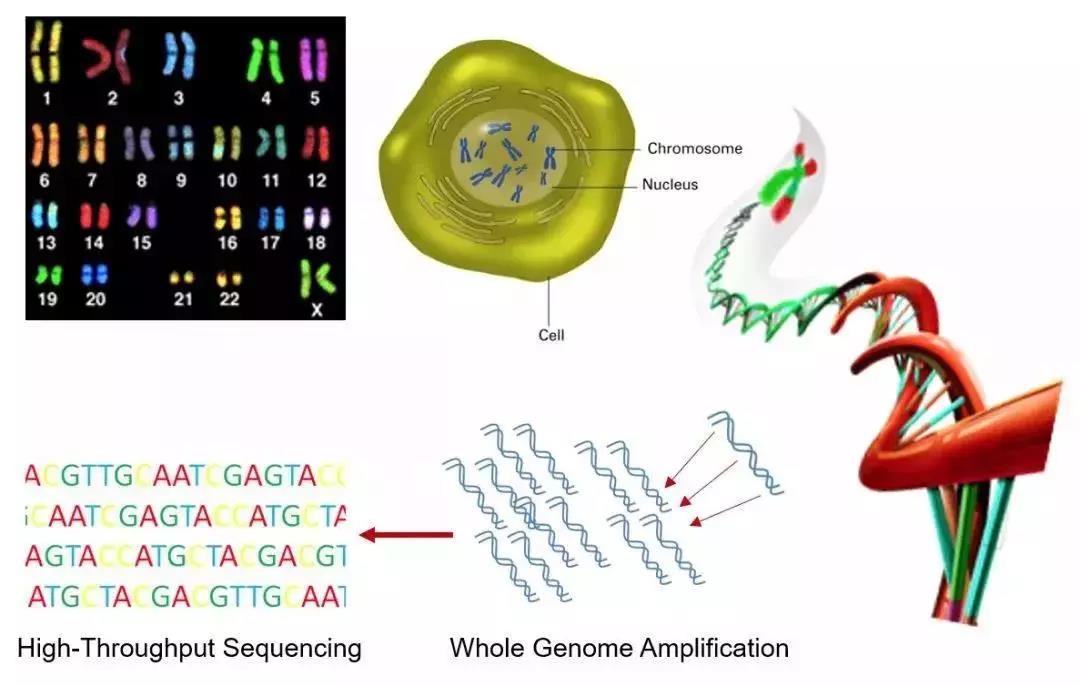
There are 46 chromosomes, i.e. 46 DNA molecules in the nucleus of a human somatic cell, 23 of which are from the father and 23 from the mother. In DNA, there are four different bases: adenine (A), cytosine (C), guanine (G) and thymine (T). Adenine pairs with thymine and guanine pairs with cytosine. One human somatic cell contains a total of 6 billion base pairs. The sequence of these base pairs encodes genetic information, comprising the human genome. Much of this sequence is identical from person to person, except for variations in 1 out of 1000 base pairs on average. Mutations in the base sequence of DNA may lead to genetic diseases or cancer. The completion of the Human Genome Project in 2001 is a milestone in the history of mankind, though the resulting genome was a mosaic of the DNA sequence of several persons instead of just one individual.
Single-cell DNA amplification and sequencing allows us to obtain the DNA sequence of all 46 chromosomes in a human cell
Not only does each person's genome differ, but the genome of each cell is also distinct, since DNA in living cells may change with time as they acquire mutations. Traditional sequencing approach lacks the precision and sensitivity to detect genetic variations of individual cells, while MALBAC allows for the low-bias whole genome amplification of single cells, so that even one mutation out of 6 billion base pairs could be discovered. In many cases, such as fertilized eggs and circulating tumor cells, just one or perhaps a few cells is at stake, making MALBAC a powerful tool in both science and medicine.
On my 50th birthday, the reunion at Harvard with Xie lab members in the span of twenty years and some of my colleagues from BIOPIC
My greatest joy at Harvard was the process of innovation, endeavoring with my students and postdocs day and night. Many amongst them are luckier than I was – they had produced important works during their Ph.D. and postdoctoral periods. I am also gratified to see them in faculty positions at over forty major universities around the globe, many recognized as experts or leaders in their own fields, such as Bob Dunn of University of Kansas, Lukas Novotny of ETH Zurich, and Andreas Zumbusch of University of Konstanz, Peter Lu of Bowling Green State University, Eric Potma of University of California at Irvine, Antoine van Oijen of University of Wollongong, Hao Yang of Princeton University, Jixin Cheng of Boston University, Peng Chen of Cornell University, Long Cai of Caltech, Nir Friedman of the Weizmann Institute of Science, Jie Xiao of Johns Hopkins University, Ji Yu of University of Connecticut, Johan Elf of Uppsala University, Guoqing Zhang of University of Science and Technology of China, Wei Min and Peter Sims of Columbia University, Conor Evans of Harvard Medical School, Will Greenleaf of Stanford University, Chenghang Zhong of Baylor College of Medicine, Paul Blainey and Jinwei Li of MIT, Xiaolin Nan of Oregon Health and Science University, Dan Fu of University of Washington, Minbiao Ji of Fudan University, Lingjie Kong of Tsinghua University, Lu Fake of New York State University, and the list goes on.
Then there were those who are able to translate the scientific contributions from our lab into commercial technologies. Among them are Sijia Lu, one of the inventors of MALBAC who returned to China after getting his Ph.D, successfully employing MALBAC in IVF to help prevent transmission of hereditary diseases, and Chris Freudiger, co-inventor of Simulated Raman Scattering microscopy to be applied in neurosurgery.
In 2009, Harvard appointed me as its Mallinckrodt Professor of Chemistry and Chemical Biology. However, in my heart, the idea of returning to where I once was had begun to emerge.
Journeying home
2018 marks the 40th anniversary of the Chinese Economic Reform. After settling down in the United States, each time I returned to China I was astounded and moved by its tremendous progress. Thanks to the Economic Reform and our present age, we are fortunate to encounter the fastest growing period in modern China. When I returned to Beijing to watch the 2008 Olympics, I was heartened by the Chinese team winning the most gold medals, but I knew we have a long way to claiming first place in the realm of science.
I became a visiting professor at the Peking University College of Chemistry and Molecular Engineering in 2001. In 2009, Rao Yi, then Dean of the PKU School of Life Sciences, persuaded me to return full time to Beida. I was appointed Cheung Kong Visiting Professor at Beida in the same year. Later, together with Professors Su Xiaodong and Huang Yanyi, both of them returnees and Beida alumni, we proposed to our alma mater to found the Biodynamic Optical Imaging Center (BIOPIC), which was met with strong support from the university leadership. BIOPIC was inaugurated in December 2010. The name "BIOPIC" stems from my earlier work in single-molecule optical imaging, and the center aims at establishing a biomedical research hub empowered by technological innovations- advance in life science is driven by breakthroughs in research tools and the cross-integration of multiple disciplines. Recently, the center has been renamed Biomedical Pioneering Innovation Center, still known as BIOPIC.
BIOPIC Inauguration Ceremony in 2010
BIOPIC has attracted an assemblage of outstanding talents from overseas. Professor Tang Fuchou was the first young returnee scholar in our center, recruited from Cambridge University. He has since become an up and coming scientific star both at home and abroad. Professor Zhang Zemin joined us from the United States as a member of the Thousand Talents Program. Eight years have passed since the founding of BIOPIC, our scientists have published many top papers, doing path-breaking research at the frontiers of life science, and making significant advances on medical applications. For several years now, I have been back and forth between Peking and Harvard University, building a close collaboration between my two lab groups in the two great institutions. Over time, BIOPIC has grown to become an international leader in the field of single-cell genomics.
BIOPIC's sequencing platform
In a joint effort with Professor Qiao Jie's group at Peking University Third Hospital, and Prof. Tang Fuchou's and my own group in BIOPIC, we used MALBAC technology to help parents who are carriers of monogenic diseases to have healthy offspring through IVF [12]. I had always wished for my work in single-molecule analyses to be used in benefiting society, and I was pleased that it was in Beida where my dream came true.
There are currently more than 6,000 known monogenic diseases. In a patient's somatic cell are two alleles of the same gene, one from the father and one from the mother, and usually, the disease causing mutation is only in one of the two alleles. As a random event involving the single molecules, the disease allele from the patient has a 50% chance of being passed to their offspring, which we used to leave up to fate! Our approach overcomes stochasticity with precision, fighting fate by using MALBAC to screen for and implant only those fertilized eggs free of the disease allele.
Visiting the first MALBAC baby with Qiao Jie (second from left) and Tang Fuchou (first from right)
I still remember how thrilled I was on September 19th, 2014, when I held the first "MALBAC baby" in my arms. This achievement epitomizes precision medicine in action. To date, the MALBAC technology has successfully helped hundreds of parents in China avoid the transmission of monogenic diseases to their offspring. I am very proud that our work in Beida was able to advance medicine and contribute to enhancing human health.
Group photo taken at the BIOPIC 2018 Annual Meeting
In 2016, with support from the Beijing municipal government, the Beijing Advanced Innovation Center for Genomics (ICG) was established in Peking University. We hope to conduct even more cutting-edge research in genomics and related fields, and that the fruits of our discovery would ultimately benefit mankind.
With members of my lab at Peking University, 2018
The 2018 graduation season is upon us. This will be the last time I attend Harvard's commencement on stage as a member of its faculty, and I am very glad that my son is part of this commencement as a graduate of Harvard College. I was also there for the high school graduation ceremony of my twin daughters, who will be leaving for college. I am grateful that my children are all grown up now, and I could afford to return to Beida to continue my research.
A recent photo with my son
As I write, the Weiming Lake Ice Arena has opened once again, and I was reminded of my schooldays when my classmates would race each other on their skates in the wintery cold. My exhilaration as I glided across the lake is still fresh upon my mind: from childhood, college, till now, ice skating and skiing are my favorite sports – Beida has endowed me with a lifelong hobby! Now, as I watch a new generation of students on ice, I couldn't help but be brought back to the 1980s, the time of my own youth – in every era, the face of the Beida student will always be the face of Peking University!
Profile
Professor Xiaoliang Sunney Xie is an internationally renowned biophysical chemist. He was born in Beijing in 1962. He received his B.Sc. in Chemistry from Peking University in 1984, and his Ph.D. in Physical Chemistry in 1990 from University of California at San Diego. He conducted postdoctoral research at the University of Chicago and in 1992 joined Pacific Northwest National Laboratory. In 1998, he became a tenured professor of the Department of Chemistry and Chemical Biology at Harvard University. From 2009 to 2018, Xie worked as the Mallinckrodt Professor of Chemistry and Chemical Biology at Harvard. Since 2010, he has been the Director of Biodynamic Optical Imaging Center (now renamed Biomedical Pioneering Innovation Center, BIOPIC) at Peking University, and in 2016 he became the Director of Beijing Advanced Innovation Center for Genomics (ICG). Starting July of 2018, he became the Lee Shau-kee Professor of Peking University.
Note: "Beida" is the Chinese abbreviation of Peking University
References:
1. Xie, X.; Simon, J. D. “Picosecond time resolved circulardichroism spectroscopy: experimental details and applications,” Rev SciInstrum 60, 2614-2627 (1989).
2. Lu, H. Peter; Xun, L.; Xie, X. Sunney "SingleMolecule Enzymatic Dynamics," Science 282, 1877 (1998).
3.Zumbusch, A.; Holtom, G. R.; Xie, X. Sunney. "Vibrational Microscopy Using Coherent Anti-Stokes Raman Scattering," Phys. Rev. Lett. 82, 4142 (1999).
4.https://harvardmagazine.com/2012/11/studying-the-stele
5.潘永祥,吴自勤,范淑兰,物理,8,493-500(1993).
6. Yu, J.; Xiao, J.; Ren, X.; Lao, K.; Xie, X. Sunney. ; "Probing Gene Expression in Live Cells, One Protein Molecule at a Time," Science, 311, 1600-1603 (2006).
7. Cai, L.; Friedman, N.; Xie, X. "Stochastic protein expression in individual cells at the single molecule level," Nature, 440, 358-362 (2006).
8. Freudiger, C. W.; Min, W.; Saar, B. G.; Lu, S.; Holtom, G. R.; He, C., Tsai, J. C.; Kang, J.; Xie, X. Sunney "Label-Free Biomedical Imaging with High Sensitivity by Stimulated Raman Scattering Microscopy" Science, 322, 1857-1861 (2008).
9. Ji, M.; Lewis, S.; Camelo-Piragua, S.; Ramkissoon, S. H.; Snuderl, M.; Venneti, S.; Fisher-Hubbard, A.; Garrard, M.; Fu, D.; Wang, A. C.; Heth, J. A.; Maher, C. O.; Sanai, N.; Johnson, T. D.; Freudiger, C. W; Sagher, O.; Xie, X and Orringer, D. A. "Detection of human brain tumor infiltration with quantitative stimulated Raman scattering microscopy," Sci Transl Med 7(309), 309ra163, DOI:10.1126/scitranslmed.aab0195 (2015).
10. Sims, P. A.; Greenleaf, W. J.; Duan, H.; Xie, X. Sunney. "Fluorogenic DNA Sequencing in PDMS Microreactors," Nat Methods 8, 575-580 (2011).
11. Zong, C.; Lu, S.; Chapman, Alec R.; Xie, X. "Genome-Wide Detection of Single-Nucleotide and Copy-Number Variations of a Single Human Cell," Science 338, 1622-1626 (2012)
12. Yan, L.; Huang, L.; Xu, L.; Huang, J.; Ma, F.; Zhu, X.; Tang, Y.; Liu, M.; Lian, Y.; Liu, P.; Li, R.; Lu, S.; Tang, F.; Qiao, J.; Xie, X. Sunney. "Live births after simultaneous avoidance of monogenic diseases and chromosome abnormality by next-generation sequencing with linkage analyses," Proc Natl Acad Sci USA, 112, 15964-15969, (2015).
Written by: Xiaoliang Sunney Xie
Translated by: Yeqin Ma, Qiu Shi Science and Technologies Foundation
Edited by: Huang Weijian
Photo credits to: Xiaoliang Sunney Xie, Zeng Liang
Source: The Charms of Spirit 2018 (First Edition, May 2018), Peking University News Center, Peking University Press